|
The problem lay in the origins of the GAMP guide. For example, when a commercially available laboratory system was validated, functional and design specifications were written for virtually no gain but at a great cost in time and resources. 
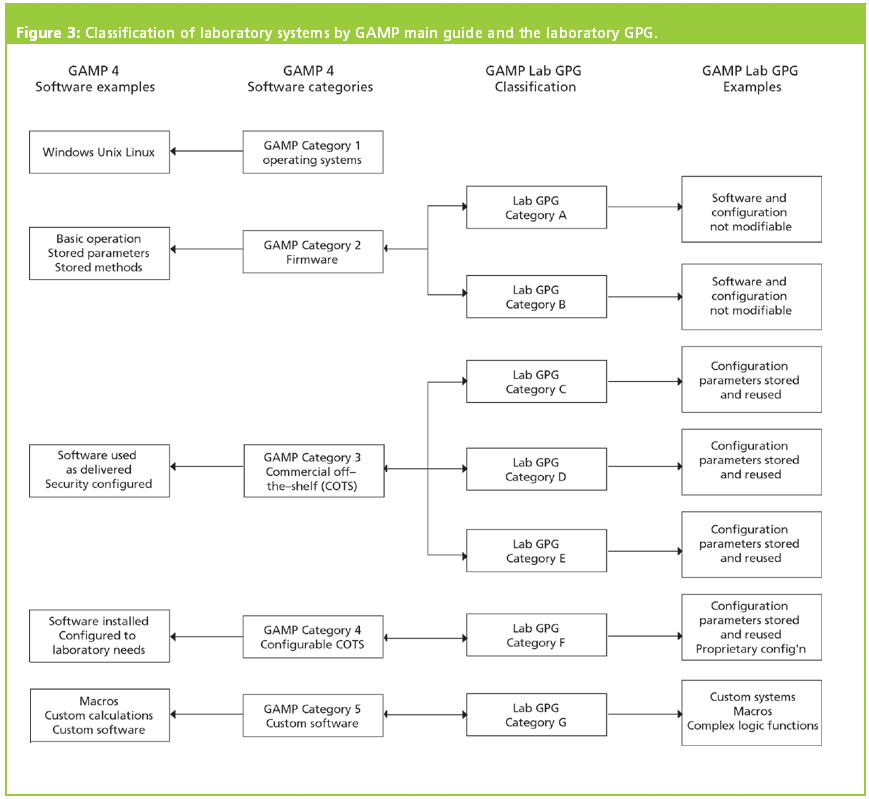
Therefore, every validation was shoehorned into an inappropriate model because there was little thought and intelligence applied and the model followed blindly. It had very little to do with computerized systems, especially laboratory ones. My rationale for this position is that versions 1–4 of the guide presented a single life cycle V model that was really only applicable to process equipment and manufacturing systems. I love the classification of software outlined in Appendix M4 and hate the life cycle V model. However, before we continue much further I should also declare a vested interest: I have a love–hate relationship with the GAMP guide. We will explore version 5 of the software classification and see what changes we need to make to ensure that it can be implemented practically and effectively in any laboratory. This is one of the best parts of the guide as it has an in-built risk assessment, as we shall see in this column. This publication has been available since 1994, when version 1 was informally published in the UK, and since its inception it has always contained a classification of software. Version 5 of the Good Automated Manufacturing Practice (GAMP) guide (1) was released last year. This column will look at what the changes mean for the laboratory and whether all of these should be implemented. 
The Guide facilitates the effective and efficient use of valuable resources by the application of appropriate and proportionate practices, encouraging innovative approaches to managing risk to patient safety, product quality, and data integrity, while supporting benefit to public health.The GAMP (Good Automated Manufacturing Practice) guide version 5 was released in March 2008 and one of the changes was that the classification of software was revised again. Technological innovation is essential for life sciences industries in providing value to society while also controlling costs and reducing time to market. The ISPE GAMP ® 5 Guide: A Risk-Based Approach to Compliant GxP Computerized Systems Second Edition aims to protect patient safety, product quality, and data integrity by facilitating and encouraging the achievement of computerized systems that are effective, reliable, and of high quality. GAMP ® guidance does not define a prescriptive method or a standard, but rather provides pragmatic guidance, approaches, and tools for the practitioner.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
